Reduce non added value compliance time & costs
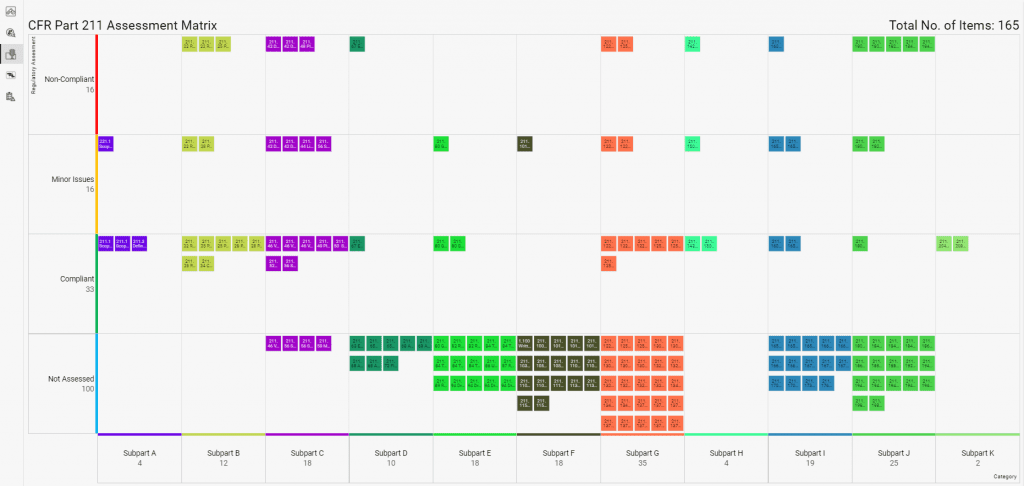
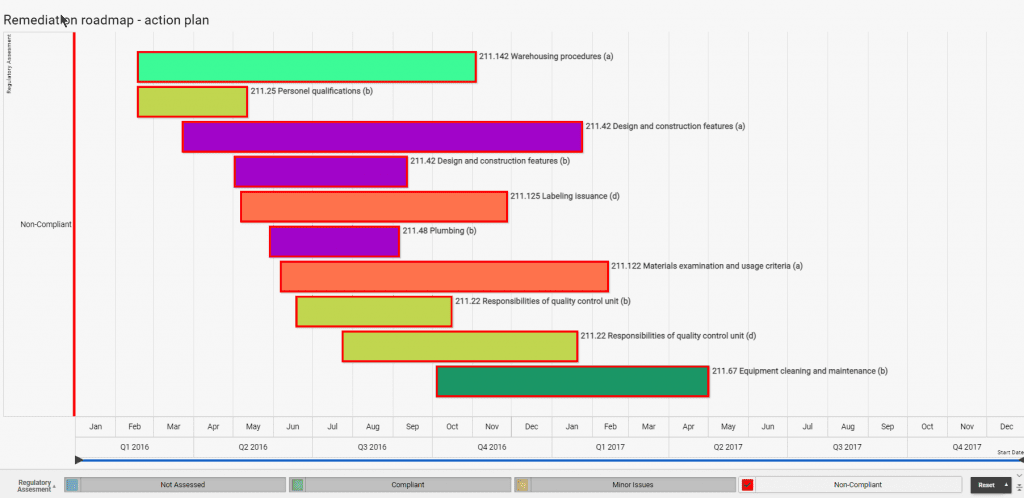
If you have to collate audit reports and information from various sites, teams or functions and consolidate this information into an overall regulatory compliance status, R-PRC can help you reduce the non-added value administration time by providing your compliance auditors and staff an online form to complete, which when submitted updates your risk based assed equipment, systems and processes.
No more updating spreadsheets, presentations or reports and emailing them. All your dashboard views and presentations are updated, ensuring everyone has the most current status to help remediate and prevent the non-compliance reoccurring within your business.