“SharpCloud is a very good tool. You can organise your work, projects, ideas, strategy, roadmap whatever your organization is working on or planning to. In organisations that are widely spread and across the globe it is the perfect collaboration tool.”
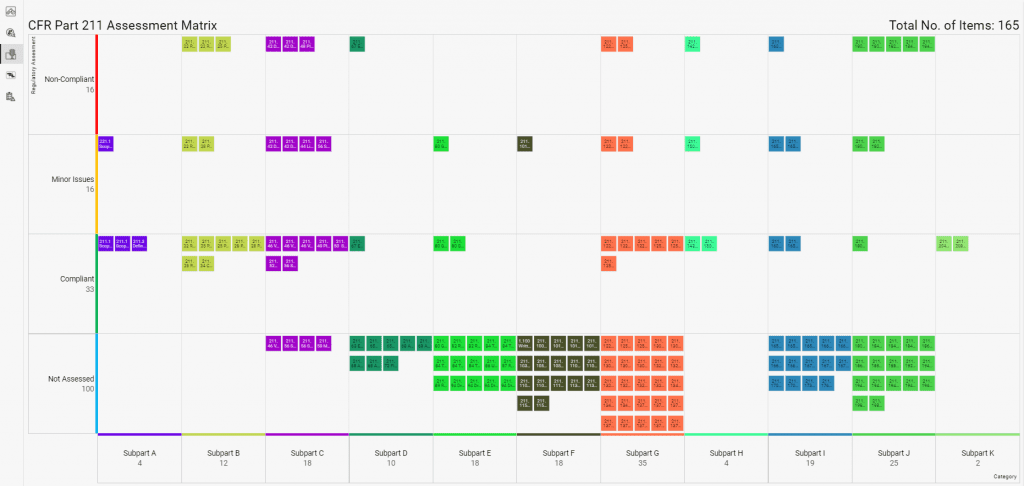
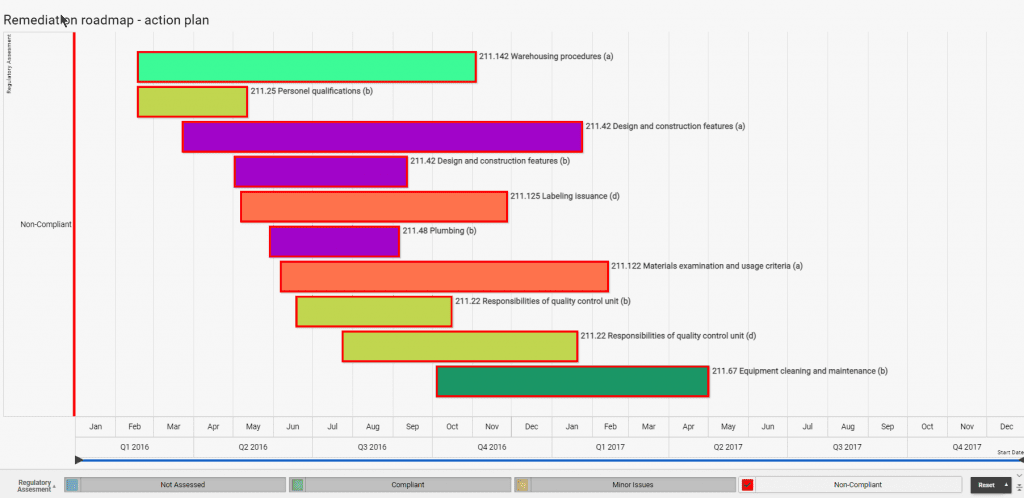
Build quality in by sharing regulatory insights and best practices with R-PRC
(Roelto – Pharmaceutical Regulatory Compliance)